First Application of an Absorbable Skin Stapler in Peripheral Vascular Surgical Procedures
Vierhout BP, de Korte JD, De Vos B, Bottema JT, Zeebregts CJ
DOI10.4172/2472-1905.100027
Vierhout BP1*, De Korte JD1, De Vos B1, Bottema JT2 and Zeebregts CJ2
1Department of Surgery, Wilhelmina Hospital, Assen, Netherlands
2Department of Surgery, Division of Vascular Surgery, University Medical Center Groningen, University of Groningen, Groningen, Netherlands
- *Corresponding Author:
- Vierhout BP
Department of Surgery
Wilhelmina Hospital, Europaweg-Zuid 1
9401 RK Assen, Netherlands
Tel: +31 592 325 443
Fax: +31 592 325 407
E-mail: bas.vierhout@wza.nl
Received Date: March 08, 2017; Accepted Date: March 23, 2017; Published Date: March 30, 2017
Citation: Vierhout BP, De Korte JD, De Vos B, et al. First Application of an Absorbable Skin Stapler in Peripheral Vascular Surgical Procedures. J Aesthet Reconstr Surg. 2017, 3:4. doi: 10.4172/2472-1905.100027
Abstract
Abstract Objective: Appropriate skin closure is essential and several techniques are available such as dermal sutures and metal staples. Absorbable staples have promising qualities. Our aim was to lower the surgical site infection (SSI) rate with the use of subcuticular absorbable skin (Insorb™) staples.
Design and Patients: A long-term (IDEAL) phase 4 historical comparative effectiveness trial was performed during a 2-year period from December 2012 to December 2014 in patients undergoing an infra-inguinal femoro-popliteal bypass operation. We compared closure of the skin by means of absorbable stapler with a cohort of patients treated with intracutaneous sutures. Primary outcome: number of SSI’s. Secondary outcome: other complications, duration of surgery and hospital length of stay.
Main Results: 44 patients were enrolled in the study; 6 patients underwent skin closure with the absorbable staples. 4 of them had an SSI. Of 38 patients treated with intracutaneous sutures 7 had an SSI (risk difference: 0.48, 95% CI 0.09-0.88). Duration of surgery (p= 0.84) and length of stay (p= 0.15) were similar.
Conclusion: Due to a large proportion of SSI’s in the absorbable stapler group the use of these subcuticular staples was discontinued in our facility. We consider the absorbable staples not strong enough for wound closure in peripheral vascular surgery.
Keywords
Absorbable skin stapler; Peripheral vascular surgery; Surgical site infection
Introduction
Appropriate surgical incision closure plays a key role in the overall outcome of each surgical procedure. Several techniques and materials have been described to achieve closure of the skin. Widely adopted are transcutaneous non-absorbable sutures, absorbable sutures and metal staples. Since 2005, a new technique was developed using subcuticular absorbable staples of polylactic/polyglycolic copolymer (comparable with braided absorbable threads such as Vicryl™), named Insorb™ Absorbable Skin Stapler (Incisive Surgical Inc., Plymouth, MN, USA) [1].
Reducing the number of SSI’s is of large interest for patients and surgeons, based both on the reduction of morbidity and costs. An increasing number of publications are available concerning wound closure with absorbable sutures. These reports show positive results regarding pain, cosmetics and absorptivity in wound closure after caesarean sections and in plastic surgery [2-4]. In vascular bypass surgery wounds are a major challenge for the surgeon; besides limb salvage and distal wound healing, wound healing of the inflicted incisions must be reached. It was anticipated that for this reason, the application of the new absorbable Insorb™ staples could positively attribute to a better wound healing. Because of these possible advantages, we decided to test Insorb™ Staples in cardiovascular compromised patients who were indicated for infra-inguinal bypass surgery in our facility. We expected the rapidly applicable absorbable staples to reduce the SSI-rate in femoro-popliteal bypass surgery. A comparison was made with a cohort of patients undergoing an infra-inguinal bypass in the preceding two years. Special attention was paid to the incidence of infection and wound healing.
Methods
Study design
The surgical procedures were performed during a 2-year period from December 2012 to December 2014 at Wilhelmina Hospital Assen, the Netherlands. The design was consistent with a phase 4 study as described in the IDEAL framework [5]. A comparison was made of absorbable skin staples with a prospectively maintained database of patients treated with intracutaneous sutures. Retrospectively, these data were reviewed for patient characteristics. Patients’ data were analyzed anonymously.
Ethical considerations
Institutional Review Board approval for the introduction of this skin closure technique was obtained. Informed consent was received for the patients prospectively included. We had no plans for publication, but because of the rare outcome this seemed mandatory post-hoc [5]. As a consequence, besides the Institutional Review Board, the Medical Ethical Commission was not informed. Study information for the patients was provided in advance during their preoperative visit. They were informed about the procedure and required compliance. All patients were given one-week time to make their decision regarding participation in the study and all gave informed consent (absorbable stapler group).
In the intracutaneous sutures group retrospective patient file research was not in the scope of the Dutch Human Bound Research and ethical approval was not required.
Study population
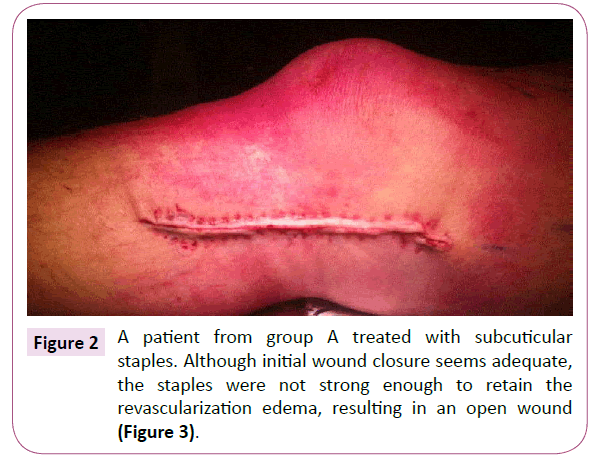
Table 1 shows the in-and exclusion criteria for enrollment in this study. The absorbable stapler group (group A) consisted of a group of prospectively screened patients that underwent infra-inguinal bypass surgery with absorbable skin staples. The staples were deployed capturing the dermal layer of both sides of the incision. The stapler contains 20 individual, horseshoe-shaped staples, 5 mm long x 3.5 mm wide x 0.7 mm thick. Closures were accomplished by having an assistant approximate and elevate the skin edges into the stapler device, using 2 forceps, while the surgeon fired the stapler (Figures 1 and 2). All involved vascular surgeons and surgical assistants were trained on the use of the dermal stapler. All had the opportunity to use the stapler in a testing setting.
| Group A: Absorbable staples (prospective) | Group B: Control cohort (retrospective) | |
|---|---|---|
| Inclusion criteria | •Age >18 years •Suitable for peripheral bypass surgery •Adequate popliteal landing zone |
|
| Exclusion criteria | •Incapacity (mentally disabled or legal disqualification) •Severe concomitant illness that drastically shortens life expectancy or increases risk of therapeutic interventions |
|
Table 1: Inclusion and exclusion criteria.
Group B, which served as a control group, consisted of a prospectively maintained database with retrospectively recorded characteristics treated with intracutaneous sutures. They underwent surgery in the time period December 2012 to July 2014, with closure of the wound using a running absorbable suture (Caprosyn™ 3-0 monofilament absorbable suture, Covidien, Mansfield, MA, USA).
Patients in both groups underwent surgery with closure of the subcutaneous tissue with a running 3-0 braided polygalactin suture (Polysorb; US Surgical, Norwalk, CT, USA).
Data collection and outcomes
Data recorded included age, sex, Fontaine classification, risk factors (smoking habits, diabetes mellitus, hypertension, hypercholesterolemia, familial predisposition), side of the operation, duration of surgery, supra- or infragenual landing zone of the bypass, autologous vein or PTFE (either heparin coated or not) and hospital length of stay. Post-procedure complications were recorded, as well as the need for redo surgery. Primary, primary assisted and secondary patencies of the bypass were calculated.
The primary outcome was the rate of Surgical Site Infections (SSI’s). Secondary outcomes were the numbers of seroma formations and rebleeding necessitating reoperation. Also the duration of operation and the hospital length of stay were evaluated, as were the patency of the bypass and the necessity of an additional amputation.
The definition of a SSI was purulent drainage from the incision, organisms isolated from an aseptically obtained culture in case the wound opened spontaneously, or an incision that was deliberately opened by a surgeon for reasons of suspicion for infection, and its culture positive or not cultured. The patient had at least one of the following signs or symptoms: pain or tenderness, localized swelling, erythema, or heat, as mentioned in the CDC-guidelines [6].
In patients treated with the absorbable stapler (group A) and patients from group B, hospital-location and treating vascular surgeons and assistants were unchanged.
Statistics
In 2015, the incidence of reported SSI’s in infra-inguinal bypass surgery was 15% in the Netherlands [7]. We hoped to reach a reduction of our infection rate from 18% (our own historical data) to 9%. A power-analysis with an effect size of 90% and a margin of 10% in the formula; n= 8 x (p1(1-p1) + p2(1-p2))/(p1-p2) [8], calculated 227 patients in each group necessary to avoid a type II error (alpha 5%, power of 80%) [8].
The proportion of patients complicated with a SSI was calculated with risk differences (RD) and their corresponding 95% confidence intervals (CI) (Standard Error (SE)=√[pA(1-pA)/nA + pB(1-pB)/nB]). When this CI includes zero, the H0 is accepted, meaning no significant difference between the proportions. The same method was used for the risk factors of the participants in each group and their secondary outcomes. Differences between continuous variables, such as age, duration of surgery, hospital length of stay and duration of follow up, were calculated using the Students t-test. Normal distribution was tested with Kolmogorov-Smirnov test and in case of skewness a log-or inverse-transformation was performed. Significance was set at p<0.05.
Results
Demographics
A total of 6 subjects with an indication for infra-inguinal bypass surgery were identified and gave permission for inclusion in the absorbable stapler group, coded as group A. Patient demographics and operative parameters are shown in Table 2. The retrospective study cohort intracutaneous sutures consisted of 38 consecutive patients, who had been treated with a running intradermal suture: group B. Patient risk factors were not significantly different between the two groups, except for familial predisposition and the number of patients with Fontaine grade III classification; they were more common in group B. The latter group also enclosed patients treated for popliteal aneurysms, two with minor symptoms of distal ischemia (Table 2).
A completion angiogram was routinely performed since the beginning of 2013. As a consequence, not all patients of group B had an angiographic evaluation at the end of the operation (16%).
| Group A: Insorb™ stapler |
Group B: Intracutaneous sutures |
||||
|---|---|---|---|---|---|
| Number of subjects | 6 | 38 | - | ||
| Mean age in years (range) | 72 (53-85) | 68 (46-90) | 68.5 (63.1-81.3) | ||
| n | pption | n | pption | RD (95%-CI) | |
| Male gender | 4 | 0.67 | 22 | 0.58 | 0.09 (-0.32-0.5) |
| Risk factors | |||||
| Smoking habits | 2 | 0.33 | 16 | 0.42 | 0.09 (-0.32-0.5) |
| Diabetes mellitus | 2 | 0.33 | 15 | 0.4 | 0.06 (-0.35-0.47) |
| Hypertension | 4 | 0.67 | 29 | 0.76 | 0.10 (-0.3-0.5) |
| Hypercholesterolemia | 4 | 0.67 | 27 | 0.71 | 0.04 (-0.36-0.45) |
| Familial predisposition | 0 | 0.0 | 13 | 0.34 | 0.34 (0.19-0.49) |
| Fontaine classification* | |||||
| I | 0 | 0.0 | 1† | 0.03 | 0.03 (-0.02-0.08) |
| IIa | 0 | 0.0 | 2 † | 0.05 | 0.05 (-0.02-0.12) |
| IIb | 2 | 0.33 | 14 | 0.37 | 0.04 (-0.37-0.44) |
| III | 0 | 0.0 | 13 | 0.34 | 0.34 (0.19-0.49) |
| IV | 4 | 0.67 | 12 | 0.32 | 0.35 (-0.05-0.76) |
| Chronic distal wounds present | 4 | 0.67 | 12 | 0.32 | 0.35 (-0.05-0.76) |
| Affected right leg | 3 | 0.5 | 20 | 0.53 | 0.03 (-0.4-0.46) |
| Mean DOS in minutes (range) | 183 (156-215) | 178 (162-195) | 179 (164-194) | ||
| Venous bypass | 6 | 1.0 | 25 | 0.66 | 0.34 (0.19-0.49) |
| Anastomosis below the knee | 4 | 0.67 | 21 | 0.55 | 0.11 (-0.29-0.52) |
| DSA at the end of the operation | 6 | 1.0 | 32 | 0.84 | 0.15 (0.04-0.27) |
Table 2: Demographics and risk factors.
Primary outcome
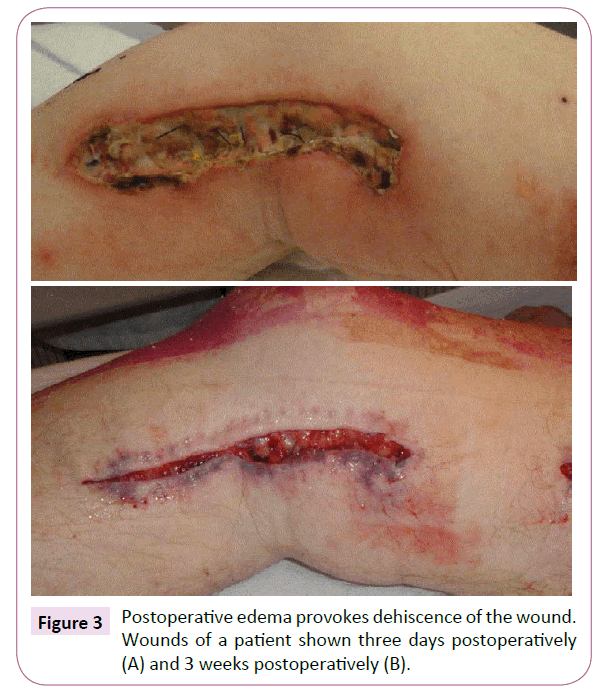
A total of 4 wounds of group A (n=6) were complicated with a SSI (67%). Three wounds opened spontaneously, due to traction on the skin. Staples were removed, but the wound was not cultured. The fourth patient had a positive culture for Pseudomonas aeruginosa. Due to extensive edema after revascularization, the staples seemed unable to contain continuity of the skin (Figures 3 and 4).
In control group B (n=38) 7 patients (18%) had a surgical site infection (SSI). The risk difference (RD) between group A and B was 0.48 in favor of group B (95% CI 0.09-0.88). Four of these 7 patients of group B needed a second operation due to this infection (Table 2). Two patients had cultures positive for Staphylococcus aureus, one contained Bacteroides fragilis, and the 4th was infected with Escherichia coli and Enterococcus faecalis. The other 3 had a minor SSI with dehiscence of the wound. One wound was cultured and showed growth of Staphylococcus aureus, the remaining 2 were not cultured.
Secondary outcomes
Postoperative seroma formation was only seen in group B (7/38= 8%) (RD 0.18, 95% CI 0.06-0.31). One of these 7 patients had a subsequent wound infection, but no organisms were identified from the cultured wound fluid. The remaining 6 wounds with a seroma healed without further complications. One patient from group A had profuse lymph leakage. The seroma drained through the staples. No SSI occurred.
Re-operation for persistent bleeding was performed in two patients from group B (RD: 0.05, 95% CI -0.02-0.12). Duration of surgery was similar in both groups: the mean of group A was 183 min (95% CI 156-215 minutes) versus a mean of group B 178 min (95% CI 162-195 min) (p=0.84, normality after log-transformation). Mean hospital length of stay for group A was 6.7 days (95% CI 4.6-12.1) and for group B 4.7 days (95% CI 4.1- 5.5) (p=0.15), with an overall mean hospital length of stay of 4.9 days (95% CI 4.2-5.7 days) (normal distribution after inverse transformation).
One patient from group A had a toe amputation due to persisting ischemia. Five patients of group B were treated with an additional amputation (forefoot-, below knee amputation, and through knee amputation). Two patients (5%) died within 30 days post-surgery, due to congestive heart failure after surgery (Table 3).
| Group A | Group B | ||||
|---|---|---|---|---|---|
| Insorb™ Stapler | Intracutaneous sutures | ||||
| Number of subjects | 6 | 38 | - | ||
| n | pption | n | pption | RD (95%-CI) | |
| Surgical site infection (SSI) | 4 | 0.67 | 7 | 0.18 | 0.48 (0.09-0.88) |
| SSI requiring re-operation | 0 | 0.0 | 4 | 0.11 | 0.11 (0.01-0.2) |
| Bleeding requiring re-operation | 0 | 0.0 | 2 | 0.05 | 0.05 (-0.02-0.12) |
| Seroma | 0 | 0.0 | 7 | 0.18 | 0.18 (0.06-0.31) |
| Toe amputation | 1 | 0.17 | 0 | 0.0 | 0.17 (-0.13-0.46) |
| Forefoot amputation | 0 | 0.0 | 2 | 0.05 | 0.05 (-0.02-0.12) |
| Leg amputation | 0 | 0.0 | 3 | 0.08 | 0.08 (-0.01-0.16) |
| Death of congestive heart failure | 0 | 0.0 | 2 | 0.05 | 0.05 (-0.02-0.12) |
| Mean HLOS in days (range) | 6.7 (4.6-12.1) | 4.7 (4.1-5.5) | 4.8 (4.3-5.7) | ||
| Mean follow up in months (range) | 7 (3-11) | 15 (0-30) | 14.5 (12.7-16.3) | ||
| Primary patency bypass | 5 | 83% | 22 | 49% | - |
| Primary assisted patency | 5 | 83% | 25 | 70% | - |
| Secondary patency | 6 | 100% | 27 | 80% | - |
| Lost to FU | 0 | 0% | 1 | 3% | - |
| Died during follow up | 0 | 0% | 4 | 10% | - |
Table 3: Postoperative complications after infra-inguinal bypass surgery. Patencies are calculated at the time of follow up (mean 7 months for group A and mean 15 months for group B).
After a mean follow up of 6.8 months all conduits were patent for group A. One patient of this group was re-operated for closure of a persisting arteriovenous fistula following an in-situ venous femoro-tibial bypass (Table 3). The mean follow up with duplex ultrasound examination of group B was 15 months and available for 31 patients (82%); 2 patients had died in the first days postoperatively (5%), 4 patients (11%) expired during follow up from conditions unrelated to infra-inguinal vascular disease, and 1 patient was lost to follow up (3%). During follow up 6 bypasses (16%) failed due to thrombosis. This occurred after a mean time of 12 months (2, 4, 10, 13, 22, and 23 months) after initial operation. The occluded bypasses were above the knee with vein in 1 and below the knee with vein in 2 patients. PTFE was used in 1 patient above the knee and 2 below the knee. One graft was successfully recanalized with intra-arterial mediated thrombolysis and additional stenting of the distal anastomosis. One patient was treated with a second open surgical bypass below the knee. Two patients were not suitable for redo surgery and an amputation was performed. The remaining 2 patients had healed wounds without complaints.
During follow up 3 other bypasses showed a proximal or in-bypass stenosis. These patients underwent an additional percutaneous intervention, one patient in the common iliac artery and 2 patients in the bypass.
The primary patency, primary assisted patency, and secondary patencies for group A were 83%, 83%, and 100%, respectively. For the control group (group B) these numbers were 49%, 70%, and 80%, respectively.
Discussion
This study shows that subcuticular absorbable skin staples in peripheral vascular surgery do not decrease the number of wound complications. The technique has positive aspects such as fast applicability and easy evacuation of wound fluids, but more dehiscence was seen in the intervention group. Postoperative swelling and edema seem to contribute in these problems. Absorbable staples are not strong enough, resulting in a higher infection-rate than the average 15% [7].
Endovascular surgery becomes increasingly popular. It enhances convalescence in peripheral arterial disease. Favorable outcomes are seen with a lower number of SSI’s [9]. However, there still is a necessity for open surgery in selected cases. In our cohort, severe signs of ischemia were seen. The majority of our patients (57%) needed a bypass below the knee joint (TASC C and D lesions) not suitable for endovascular techniques. And the average age of the patients was 70 years, with skin thinning.
In these vulnerable patients, we hoped to reduce wound complications with the relatively new technique. These supposed advantages are highlighted hereunder:
1. The applicability seems to be more rapid compared to dermal sutures [3,10-12]. Despite training of our vascular team, we did not find a shorter OR-duration in our intervention group. But, these were 6 venous bypasses, necessitating harvesting of the greater saphenous vein, which may explain the lack of time reduction.
2. Evacuation of excessive fluids would be an advantage of the new staples thus evacuating the postoperative seroma. One patient suffered from excessive drainage of fluids after using absorbable staples. Formation of a huge seroma was prevented. This would not have been possible in case of intracutaneous running sutures, closing the wound airtight. In the outcome, more seroma formation was seen in the control intracutaneous sutures group.
3. Metal staples require an open connection between the subcutaneous tissue and the dressings, draped upon the skin. They give more erythema and drainage [13]. Metal staples cause skin irritation and require removal [12,14]. The puncture holes result in scarring, as well [1]. Although absorbable staples seem to cause some short-term patient discomfort they also proved to have advantages over metal staples [14].
When these rapid absorbable staples do evacuate excessive wound fluid, and do not increase inflammation, we could hypothesize that only the strength of the staples was insufficient. Research shows them unsupportive after 3 weeks (<0.3 lbf*) [13]. Especially patients with skin thinning, such as elderly, are inappropriate candidates [10]. The strength was a problem larger than the drainage of fluids.
Stress on the wound edges was mentioned before to cause problems [3,11]. Additional subcutaneous sutures may prevent disruption [11], and one study even mentioned over sewing absorbable staples with extra sutures [14]. When looking at our outcomes, the number of SSI’s was higher in the absorbable stapler group compared to the sutures group (respectively 67% versus 18%). All wounds opened were classified as infected, but the number of reoperations was significantly lower than in the intracutaneous sutures group (57% of the infected wounds). These surgical sites were infected in a deeper layer. At skin-level the absorbable staples do not increase the amount of fibroblast proliferation, inflammation, or collagen deposition compared to dermal sutures [10]. SSI’s were not caused by the absorbable staples, but infection followed the dehiscence of the wound.
Limitations to the present study are numerous. The design is not randomized; numbers are small; a historic cohort was compared to a small number of prospective participants. Not all wounds were cultured when opened and duration of follow up was different between the two groups. But disappointing results urged us to warn fellow surgeons [15].
In peripheral vascular surgery wounds have a high stress on skin edges and regarding our results, we would advise against the absorbable staples in peripheral vascular surgery. Wounds in other parts of the body, e.g. in breast surgery, transplantation surgery, and facial surgery may have different characteristics and therefore less dehiscence. *lbf=pound-force (1 lbf=0.45 kg × 9.8 m/s2=4.4 Newton)
Conclusion
Although this study is underpowered, we describe the first application of an absorbable skin closure technique in major peripheral vascular procedures. Absorbable staples were compared to standard closure with running intracutaneous absorbable sutures. Because of insufficient strength of the staples and postoperative edema in peripheral vascular surgery, we advise against absorbable staples in peripheral vascular surgery.
Conflict of Interest
None of the authors declare to have a conflict of interest.
References
- Pinneros-Fernandez A, Salopek LS, Rodeheaver PF, Drake DB, Edlich RF, et al. (2006) A revolutionary advance in skin closure compared to current methods. J Long-Term Eff Med Impl 16: 19-27.
- Feese CA, Johnson S, Jones E, Lambers DS (2013) A randomized trial comparing metallic and absorbable staples for closure of a Pfannenstiel incision for cesarean delivery. Am J Obstet Gynecol 209: 551-555.
- Duteille F, Rouif M, Alfandari B, Andreoletti JB, Sinna R, et al. (2013) Reduction of skin closure time without loss of healing quality: a multicentre prospective study in 100 patients comparing the use of insorb absorbable staples with absorbable thread for dermal suture. Surg Innov 20: 70-73.
- Nitsche J, Howell C, Howell T (2012) Skin closure with subcuticular absorbable staples after caesarean section is associated with decreased analgesic use. Arch Gynecol Obstet 285: 979-983.
- McCulloch P, Altman DG, Campbell WB, Flum DR, Glasziou P, et al (2009) No surgical innovation without evaluation: the IDEAL recommendations. The Lancet 374: 1105-1112.
- Centers for Disease Control (CDC) (2015) Surgical Site Infection (SSI) event.
- https://www.volksgezondheidenzorg.info/onderwerp/zorginfecties/cijfers-context/incidentie-en-prevalentie
- Campbell MJ, Swinscow TDV (2009) Statistics at square one (11th edn.). West Sussex: Wiley-Blackwell.
- Lensvelt MM, Fritschy WM, van Oostayen JA, Holewijn S, Zeebregts CJ, et al. (2012) Results of heparin-bonded ePTFE-covered stents for chronic occlusive superficial femoral artery disease. J Vasc Surg 56: 118-125.
- Cross KJ, Teo EH, Wong SL, Lambe JS, Rohde CH, et al. (2009) The absorbable dermal staple device: A faster, more cost-effective method for incisional closure. Plast Reconstr Surg 124: 156-162.
- Tellis VA (2007) Renal transplant incision closure using new absorbable subcuticular staple device. Clin Transplant 21: 410-412.
- Broin ES, Donnell M, Donovan D, Tiernan E, Lawlor DL, et al. (1996) Absorbable skin graft staples: a clinical trial using Graftac-X. Br J Plast Surg 49: 485-487.
- Fisher DA, Bengero LL, Clapp BC, Burgess M (2010) A randomized, prospective study of total hip wound closure with resorbable subcuticular staples. Othopedics 33: 665.
- Dresner HS, Hilger PA (2009) Comparison of incision closures with subcuticular and percutaneous staples. Arch Facial Plast Surg 11: 320-326.
- (2015) Study of patients with critical limb ischemia. JACC Cardiovasc Interv 8: 1493-1502.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences